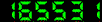MARC details
| 000 -LEADER |
|---|
| fixed length control field |
05282cam a2200253 a 4500 |
| 001 - CONTROL NUMBER |
|---|
| control field |
1844 |
| 005 - DATE AND TIME OF LATEST TRANSACTION |
|---|
| control field |
20200702113703.0 |
| 008 - FIXED-LENGTH DATA ELEMENTS--GENERAL INFORMATION |
|---|
| fixed length control field |
070410s2008 flua 001 0 eng |
| 020 ## - INTERNATIONAL STANDARD BOOK NUMBER |
|---|
| International Standard Book Number |
9780849339349 (acidfree paper) |
| 020 ## - INTERNATIONAL STANDARD BOOK NUMBER |
|---|
| International Standard Book Number |
0849339340 (acidfree paper) |
| 040 ## - CATALOGING SOURCE |
|---|
| Transcribing agency |
DLC |
| 082 00 - DEWEY DECIMAL CLASSIFICATION NUMBER |
|---|
| Classification number |
550.154 |
| Edition number |
22 |
| Item number |
D8771 |
| 100 1# - MAIN ENTRY--PERSONAL NAME |
|---|
| Personal name |
Duke, Catherine V. A. |
| 245 10 - TITLE STATEMENT |
|---|
| Title |
Chemistry for environmental and Earth sciences / |
| Statement of responsibility, etc |
Catherine V.A. Duke, Craig D. Williams. |
| 260 ## - PUBLICATION, DISTRIBUTION, ETC. (IMPRINT) |
|---|
| Place of publication, distribution, etc |
Boca Raton : |
| Name of publisher, distributor, etc |
CRC Press, |
| Date of publication, distribution, etc |
2008 |
| 300 ## - PHYSICAL DESCRIPTION |
|---|
| Extent |
230 p. |
| Other physical details |
ill. ; |
| Dimensions |
24 cm. |
| 500 ## - GENERAL NOTE |
|---|
| General note |
Includes index. |
| 650 #0 - SUBJECT ADDED ENTRY--TOPICAL TERM |
|---|
| Topical term or geographic name as entry element |
Chemistry, Physical and theoretical |
| General subdivision |
Environmental aspects |
| 650 #0 - SUBJECT ADDED ENTRY--TOPICAL TERM |
|---|
| Topical term or geographic name as entry element |
Geochemistry |
| 650 #0 - SUBJECT ADDED ENTRY--TOPICAL TERM |
|---|
| Topical term or geographic name as entry element |
Environmental chemistry |
| 700 1# - ADDED ENTRY--PERSONAL NAME |
|---|
| Personal name |
Williams, C. D. |
| 942 ## - ADDED ENTRY ELEMENTS (KOHA) |
|---|
| Koha item type |
Books |
| 505 0# - FORMATTED CONTENTS NOTE |
|---|
| Formatted contents note |
Table of Contents<br/>Preface<br/>Acknowledgements<br/>Author Biographies<br/>Chapter 1<br/>Fire<br/>Introduction to Chapter 1<br/>1.1 Atoms and Elements<br/>1.1.1 The Structure of Atoms<br/>1.1.2 The Origin of the Elements<br/>1.1.3 The Periodic Table<br/>1.1.4 Electrons and Electron Orbitals<br/>1.1.5 Radioactivity<br/>1.1.6 Radiometric Dating Methods<br/>Box 1.1 Scientific Notation<br/>1.2. States of Matter<br/>1.2.1 Plasma<br/>1.2.2 Gases<br/>1.2.3 Liquids<br/>1.2.4 Solids<br/>1.2.5 Phase Transitions and Phase Diagrams<br/>1.2.6 Pure Substances, Compounds and Mixtures<br/>1.3 Units of Measurement<br/>1.3.1 SI and Non-SI Units<br/>1.3.2 Scientific Notation and SI Prefixes<br/>1.3.3 Concentrations and Solutions<br/>Box 1.2 Significant Figures, Rounding Up and Decimal Places<br/>1.4 Chemical Bonding<br/>1.4.1 Covalent Bonding<br/>1.4.2 Cations, Anions and Ionic Bonding<br/>1.4.3 Metallic Bonding<br/>1.4.4 Electronegativity, Polar Bonds and Hydrogen Bonding<br/>1.5 Chemical Structures<br/>1.5.1 Structures of Organic Molecules<br/>1.5.2 Three Dimensional Structures, Chirality and Optical Isomers<br/>1.5.3 Structures of Inorganic Molecules<br/>1.5.4 Structures of Extended Networks<br/>1.6 Chemical Reactions<br/>1.6.1 Chemical Reactions<br/>1.6.2 The Energy of Chemical Reactions<br/>1.6.3 Chemical Equlibria and Le Chatelier's Principle<br/>1.7 Summary of Chapter 1<br/>End of Chapter Questions<br/>Chapter 2<br/>Earth<br/>Introduction to Chapter 2<br/>2.1 Formation of the Earth<br/>2.1.1 The Structure of the Earth<br/>Box 2.1 Dating the Earth<br/>2.2 The Structures of Silicate Minerals<br/>2.2.1 Silicates formed from Isolated Tetrahedra - Orthosilicates<br/>2.2.2 Single Chain Silicates - Pyroxenes<br/>2.2.3 Double Chain Silicates - Amphiboles<br/>2.2.4 Sheet Silicates - Micas, Clays and Talc<br/>2.2.5 Framework or Tecto-Silicates - Silica, Feldspars and Zeolites<br/>2.3 Igneous Rocks<br/>2.3.1 The Composition of Igneous Rocks<br/>2.3.2 Crystallisation of Igneous Rocks<br/>2.3.3 Phase Diagrams<br/>2.3.4 Trace Elements in Igneous Rocks <br/>2.3.5 Mineral Stability<br/>Box 2.2 Determining the Elemental Composition of Rocks<br/>2.4 Sedimentary Rocks<br/>2.4.1 Siliciclastic Rocks - Sandstones, clays, mudstones & siltstones<br/>2.4.2 Carbonates<br/>2.4.3 Evaporites<br/>2.4.4 The Mineral Composition of Sedimentary Rocks<br/>2.4.5 The Chemical Composition of Sedimentary Rocks<br/>2.5 Metamorphic Rocks<br/>2.5.1 Metamorphism by Recrystallisation<br/>2.5.2 Metamorphism and Chemical Reactions<br/>Box 2.3 Formation of Fossil Fuels<br/>2.6 Weathering<br/>2.6.1 Physical Weathering<br/>2.6.2 Chemical Weathering<br/>2.6.3 Biological Weathering<br/>2.7 The Chemistry of Soil<br/>2.7.1 Soil Organic Matter<br/>2.7.2 Ion exchange and Soil ph<br/>2.7.3 Soil Pollution<br/>Box 2.4 The Analysis of Polluted Soils<br/>2.8 Summary of Chapter 2<br/>End of Chapter Questions<br/>Chapter 3<br/>Water<br/>Introduction to Chapter 3<br/>3.1 The Properties of Water<br/>3.1.1 The Phase Diagram of Water<br/>3.1.2 Water and Hydrogen Bonding<br/>3.1.3 Water and Heat<br/>3.1.4 Water as a Solvent<br/>3.1.5 The Water Cycle<br/>3.2 Acids, Bases and the ph Scale<br/>3.2.1 Acids and Bases<br/>3.2.2 The Relative Strength of Acids and Bases<br/>3.2.3 Strong Acids and Bases<br/>3.2.4 Weak Acids and Bases<br/>3.2.5 The Self Ionisation of Water<br/>3.2.6 The ph Scale<br/>3.2.7 Acid-Base Titrations<br/>3.2.8 Buffer Solutions<br/>Box 3.1 The Effects of Acid Rain<br/>3.3 Ions in Solution<br/>3.3.1 The Solvation of Ions<br/>3.3.2 Sparingly Soluble Salts and Solubility Products<br/>3.3.3 The Carbonate System<br/>3.3.4 Hardness of Water<br/>3.3.5 The Chemistry of Sea Water<br/>Box 3.2 CO2 and Killer Lakes<br/>Box 3.3 Water Softeners and How They Work<br/>3.4 Redox Chemistry<br/>3.4.1 Oxidation, Reduction and Oxidation States<br/>3.4.2 Redox Potentials and Stability Field Diagrams<br/>3.4.3 Speciation<br/>3.4.4 The Redox Chemistry of Nitrogen<br/>Box 3.4 Acidic Mine Drainage<br/>3.5 Colloids and Suspended Particles<br/>3.6 Water Pollution<br/>3.6.1 Heavy Metals<br/>3.6.2 Nutrients and Eutrophication<br/>3.6.3 Organic Pollutants<br/>3.6.5 Marine oil Pollution<br/>Box 3.5 Analysis of Metals in Aqueous Solutions<br/>3.7 Summary of Chapter 3<br/>End of Chapter Questions<br/>Chapter 4<br/>Air<br/>Introduction to Chapter 4<br/>4.1 The Structure of the Atmosphere<br/>Box 4.1 Auroras<br/>4.2 Evolution and Composition of the Atmosphere<br/>Box 4.2 Formation of The Molecules of Life<br/>4.3 Natural Cycles<br/>4.3.1 The Carbon Cycle<br/>4.3.2 The Nitrogen Cycle<br/>4.3.3 The Sulfur Cycle<br/>4.4 Global Warming and the Greenhouse Effect<br/>4.5 The Ozone Layer<br/>4.5.1 Ozone Formation and UV Protection<br/>4.5.2 Ozone depletion<br/>4.6 Air Pollution<br/>4.6.1 The Key Pollutants<br/>4.6.2 Urban Smog<br/>4.6.3 Dispersal of Pollutants<br/>4.6.4 Indoor Air Quality<br/>4.7 Summary of Chapter 4<br/>Answers to Self Assessment Questions<br/> |